Oracle E-Record Events in Quality Management
OPM Quality Management supports the acquisition of electronic signatures on electronic records. Electronic documentation replaces paper with a seamless interface of business activities that improves manufacturing throughput and enhances the overall quality of operations in the supply chain. The following provides information for online and deferred events that are e-record and e-signature enabled in the OPM Quality Management application. A brief discussion of each event is provided. Refer to the Oracle E-Records Implementation Guide for details on setting up the events and approvals
This appendix covers the following topics:
- Events
- Subscriptions
- Approval Matrix
- Approval of Quality Specification
- Samples Creation
- Results
- Sample and Sample Group Disposition
- Stability Study Change Status
Events
The following table lists the events that are e-record and e-signature enabled for OPM Quality Management:
| Event | Online or Deferred |
|---|---|
| Process Quality Specifications Event | Deferred |
| Process Quality Inventory Validity Rule Specifications Event | Deferred |
| Process Quality WIP Validity Rule Specifications Event | Deferred |
| Process Quality Customer Validity Rule Specifications Event | Deferred |
| Process Quality Supplier Validity Rule Specifications Event | Deferred |
| Process Quality Monitoring Validity Rule Specifications Event | Deferred |
| Process Quality Sample Creation Event | Online |
| Process Quality Results Entry Event | Online |
| Process Quality Results Evaluation | Online |
| Process Quality Sample Disposition | Online |
| Process Quality Sample Event Disposition | Online |
| Process Quality Sample Event Disposition Composite | Online |
| Process Quality Stability Study Change Status | Online |
Events
| Name | Display Name | Description | Status | Owner Name | Owner Tag |
|---|---|---|---|---|---|
| oracle.apps.gmd.qm.spec | GMD ERES Specifications Event | ERES Event for Specifications in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.spec.vr.inv | GMD ERES Inventory Validity Rule Specifications Event | ERES Event for Inventory Specifications Validity Rule in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.spec.vr.wip | GMD ERES WIP Validity Rule Specifications Event | ERES Event for WIP Specifications Validity Rule in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.spec.vr.cus | GMD ERES Customer Validity Rule Specifications Event | ERES Event for Customer Specifications Validity Rule in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.spec.vr.sup | GMD ERES Supplier Validity Rule Specifications Event | ERES Event for Supplier Specifications Validity Rule in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.spec.vr.mon | GMD ERES Monitoring Validity Rule | ERES Event for Monitoring Validity Rule | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.spec.vr.calib | GMD ERES Calibration Validity Rule | ERES Event for Calibration Validity Rule | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.smpl.crea | GMD ERES Sample Creation Event | ERES Event for Sample Creation in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.rslt.entry | GMD ERES Results Event | ERES Event for Results Entry in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.rslt.mentry | GMD ERES Mass Results Event | ERES Event for Mass Results Entry in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.rslt.eval | GMD ERES Results Evaluation | ERES Event for Results Evaluation in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.smpl.disp | GMD ERES Sample Disposition | ERES Event for Sample Disposition in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.evt.disp | GMD ERES Sample Group Disposition | ERES Event for Sample Disposition in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.evt.dispcom | GMD ERES Sample Group Disposition Composite | ERES Event for Sample Composite Disposition in OPM Quality | Enabled | Oracle Process Manufacturing Product Development | GMD |
| oracle.apps.gmd.qm.ss.ecsts | GMD ERES Stability Study Change Status | ERES Event for Stability Study Change Status | Enabled | Oracle Process Manufacturing Product Development | GMD |
Note: It is recommended that the user entering the mass results data be the one signing for the event. In order to accomplish this, the following AME attributes must have the appropriate values:
Note: ALLOW_REQUESTOR_APPROVAL : true
Note: ALLOW_EMPTY_APPROVAL_GROUPS: true
Note: TRANSACTION_REQUESTOR_USER_ID : select fnd_global.user_id from fnd_dual
Note: Create a rule without any constraints attached to a empty approval Group.
Event Key
| Event Name | Event Key |
|---|---|
| oracle.apps.gmd.qm.spec | Spec_id |
| oracle.apps.gmd.qm.spec.vr.inv | Spec_vr_id |
| oracle.apps.gmd.qm.spec.vr.wip | Spec_vr_id |
| oracle.apps.gmd.qm.spec.vr.cus | Spec_vr_id |
| oracle.apps.gmd.qm.spec.vr.sup | Spec_vr_id |
| oracle.apps.gmd.qm.spec.vr.mon | Spec_vr_id |
| oracle.apps.gmd.qm.spec.vr.calib | Spec_vr_id |
| oracle.apps.gmd.qm.rslt.entry | Sample_id-update_instance_id-spec_id |
| oracle.apps.gmd.qm.rslt.mentry | Sample_id-update_instance_id-spec_id |
| oracle.apps.gmd.qm.rslt.eval | Sample_id-update_instance_id-spec_id |
| oracle.apps.gmd.qm.evt.disp | Event_spec_disp_id |
| oracle.apps.gmd.qm.evt.dispcom | Event_spec_disp_id |
| oracle.apps.gmd.qm.smpl.crea | Sample_id |
| oracle.apps.gmd.qm.smpl.disp | Event_spec_disp_id-sample_id |
| oracle.apps.gmd.qm.ss.ecsts | SS_Id |
Subscriptions
| Source Type | Event Filter | Phase | Status | Rule Data | Rule Function | Priority |
|---|---|---|---|---|---|---|
| Local | oracle.apps.gmd.qm.spec | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.spec.vr.inv | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.spec.vr.wip | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.spec.vr.cus | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.spec.vr.sup | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.spec.vr.mon | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.spec.vr.calib | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.rslt.entry | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.rslt.mentry | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.rslt.eval | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.smpl.disp | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.smpl.crea | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.evt.disp | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.evt.dispcom | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
| Local | oracle.apps.gmd.qm.ss.ecsts | 0 | Disabled | Key | EDR_UTIL.WF_RULE_FUNCTION | Normal |
Approval Matrix
| Oracle Application | Transaction Type ID | Transaction Type Description | Line Item Id Query String |
|---|---|---|---|
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.spec | GMD ERES Specifications Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.spec.vr.inv | GMD ERES Inventory Validity Rule Specifications Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.spec.vr.wip | GMD ERES WIP Validity Rule Specifications Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.spec.vr.cus | GMD ERES Customer Validity Rule Specifications Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.spec.vr.sup | GMD ERES Supplier Validity Rule Specifications Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.spec.vr.mon | GMD ERES Monitoring Validity Rule Specifications Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.spec.vr.calib | GMD ERES Calibration Validity Rule Specifications Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.smpl.crea | GMD ERES Sample Creation Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.rslt.mentry | GMD ERES Mass Results Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.rslt.entry | GMD ERES Results Event | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.rslt.eval | GMD ERES Results Evaluation | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.smpl.disp | GMD ERES Sample Disposition | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.evt.disp | GMD ERES Sample Group Disposition | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.evt.dispcom | GMD ERES Sample Group Disposition Composite | N/A |
| Oracle Process Manufacturing Product Development | oracle.apps.gmd.qm.ss.ecsts | GMD ERES Stability Study Change Status | N/A |
Transaction Attributes
Approval attributes are seeded for these events. You can define the attributes and use them in the rules as per the implementation instructions.
Define Rules/Associate Approvers/Associate E-Record Output XSL
The rules basing the status on Approved For Laboratory Use and Approved For General Use are seeded.
As part of the implementation, you must complete these activities. The following mandatory and non mandatory attributes must be added for all transaction types. Substitute the values shown with appropriate transaction specific values or data:
Mandatory Attributes
| Name | Attribute Type | Description | Static Usage | Usage |
|---|---|---|---|---|
| ALLOW_DELETING_RULE_GENERATED_APPROVERS | boolean | Whether to let the calling application (or its end users) delete approvers generated by the rules. | Yes | False |
| ALLOW_REQUESTOR_APPROVAL | boolean | Whether to allow requestors to approve their own transactions (when the rules do so). | Yes | False |
| AT_LEAST_ONE_RULE_MUST_APPLY | boolean | Whether to require that at least one rule apply to each transaction. | Yes | False |
| EFFECTIVE_RULE_DATE | date | The date that determines which rules are active. | Yes | (blank) |
| TRANSACTION_DATE | date | Date transaction occurred. | No | Select ame_util.versionDateToString(CREATION_DATE) from gme_batch_header where batch_id=:transactionId |
| TRANSACTION_GROUP_ID | number | Business-group ID in which transaction occurred. | Yes | (blank) |
| TRANSACTION_ORG_ID | number | Org ID in which transaction occurred. | Yes | (blank) |
| TRANSACTION_REQUESTOR_PERSON_ID | number | Person ID of person initiating transaction. | Yes | (blank) |
| TRANSACTION_REQUESTOR_USER_ID | number | User ID of person initiating transaction. | No | Select Last_updated_by from gme_batch_header where batch_id=:transactionId |
| TRANSACTION_SET_OF_BOOKS_ID | number | Set of books ID in which transaction occurred. | No | (blank) |
| USE_RESTRICTIVE_LINE_ITEM_EVALUATION | boolean | Whether to require that the same line item satisfy all line-item conditions in a given rule. | Yes | False |
| WORKFLOW_ITEM_KEY | string | The transaction's workflow item key. | No | (blank) |
| WORKFLOW_ITEM_TYPE | string | The transaction's Workflow item type. | No | (blank) |
| ALLOW_DELETING_RULE_GENERATED_APPROVERS | boolean | Whether to let the calling application (or its end users) delete approvers generated by the rules. | Yes | False |
Approval of Quality Specification
Making a Quality Specification active consists of two separate events: the approval of a specification and the approval of the specification validity rule. Process Quality Inventory, WIP, Customer, and Supplier Specification Validity Rule Events apply to item specifications. The Process Quality Monitoring Validity Rule Specifications Event only applies to the monitoring specification. Both events require the capture of e-records and e-signatures.
A specification status can be controlled through a workflow. Therefore, signatures for both creation and updates of specifications are captured deferred (electronic notifications are sent).
E-record for Specification Status Change Approval
Data on the signoff for specifications can include:
-
Spec, Description, Spec Version, Spec Status, Spec Type, Overlay, Base Spec, Base Spec Version, Owner Organization, Owner
-
Item, Item Revision, Item Description, Grade
-
Tests Summary
Seq, Test, Test Method, Replicate, Unit, Target, Min, Max
-
Tests Details
Test Seq, Test Class, Test, Test Description, Test Method, Test Data Type, From Base, Exclude, Modify, Spec Target, Spec Min, Spec Max, Test Min, Test Max, Test Unit, Expression, Test Qty, Test Qty UOM, Test Replicate, Test Priority, Optional Test, Lot Retest/Expiry Test, Out-of-Spec Action, Out-of-Spec Action Description, Out-of-Spec Action Interval, Calculate UOM Conversion, From UOM, To UOM, Experimental Error Type, Below Min, Below Min Action, Above Min, Above Min Action, Below Max, Below Max Action, Above Max, Above Max Action, Test Alias, Print Specification, Print Result, Display Decimal Precision, Report Decimal Precision
-
Spec Descriptions
Language, Description
E-record for Customer Validity Rule Status Change Approval
Data on the signoff can include:
-
Spec, Spec Version, Spec Status, Organization
-
Item, Item Revision, Item Description, Grade
-
Validity Rule Status, Customer, Operating Unit, Ship To Location
Order Number, Order Type, Order Line
-
Effective From Date, Effective To Date, Certificate Type, Required at Shipment, Required at Invoice, Required at Supplier, Lot Optional on Sample
E-record for Supplier Validity Rule Status Change Approval
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.spec.vr.sup
2. Create AME Rule for the following transaction type:
-
GMD ERES Supplier Validity Rule
Data on the signoff can include:
-
Spec, Spec Version, Spec Status
-
Item, Item Revision, Item Description, Grade
-
Organization, Internal Order, Supplier, Supplier Name, Operating Unit, Validity Rule Status
-
Site, PO Number, PO Line, Source Organization, Source Subinventory, Requisition Number, Requisition Line Number, Effective From Date, Effective To Date, Certificate Type, Required from Shipment, Required at Invoice, Required from Supplier, Lot Optional on Sample, Delayed Lot Entry
-
Create Samples Automatically, Update Inventory Quantity, Control Lot Attributes, In-Spec, Out-of-Spec
The following graphic illustrates an example of supplier validity rule status change approval event.

E-record for Inventory Validity Rule Status Change Approval
Data on the signoff can include:
-
Spec, Spec Version, Spec Status
-
Item, Item Revision, Item Description, Grade
-
Organization, Validity Rule Status, Parent Lot, Lot, Subinventory, Locator
-
Effective From Date, Effective To Date, Certificate Type, Required at Shipment, Required at Invoice, Required from Supplier, Lot Optional on Sample, Delayed Lot Entry
-
Create Samples Automatically, Update Inventory Qty, Control Lot Attributes, In-Spec, Out-of-Spec
E-record for WIP Validity Rule Status Change Approval
Data on the signoff can include:
-
Spec, Spec Version, Spec Status
-
Item, Item Revision, Item Description, Grade
-
Organization, Validity Rule Status, Batch, Recipe, Formula, Formula Line, Routing, Step, Operation
-
Effective From Date, Effective To Date, Certificate Type, Required at Shipment, Required at Invoice, Required from Supplier, Lot Optional on Sample, Delayed Lot Entry
-
Create Samples Automatically, Update Inventory Qty, Control Batch Step Status, Control Lot Attributes, In-Spec, Out-of-Spec
E-record for Monitoring Specification Validity Rule Status Change Approval
Data on the signoff can include:
-
Spec Name, Spec Version, Spec Status, Rule Type
-
Validity Rule Status, Organization, Resource, Resource Instance
-
Subinventory, Locator
-
Effective From Date, Effective To Date
E-record for Calibration Specification Validity Rule Status Change Approval
Data on the signoff can include:
-
Spec Name, Spec Version, Spec Status
-
Validity Rule Status, Resource, Resource Organization, Resource Instance
-
Start Date, End Date
Samples Creation
The Samples Creation e-record is sent when you try to approve a sample. The e-record provides all information about sample identification, including its priority, source, quality laboratory, sample dates, the specification tied to it, and any sampling plan information.
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.smpl.crea
2. Create AME Rule for the following transaction type:
-
GMD ERES Sample Creation
E-record for Sample Creation
Data on the signoff for samples creation can include:
-
Sample Number, Description, Disposition, Retain As, Priority
-
Item, Item Revision, Item Description, Source, Source Comments
-
Parent Lot, Lot, Subinventory, Locator
-
Customer, Operating Unit, Ship To Location, Order Number, Order Type, Order Line
-
Batch, Recipe, Version, Formula, Version Formula Line, Routing, Version, Step, Charge, Operation Version
-
Internal Order, Supplier, Supplier Name, Site, PO Number, PO Line, Receipt Number, Receipt Line, Source Organization, Source Subinventory, Requisition Number, Requisition Line
-
Quality Laboratory, Date Drawn, Expiration Date, Lot Retest Sample, Update Inventory Quantity, Sample Qty, Sample Qty UOM, Storage Subinventory, Storage Locator, Samples Occurrence, Specification Count, Spec Name, Spec Version, Grade
The following graphic illustrates an example of sample creation event.

Results
Quality results entry and evaluation can also require e-signature and e-record support. After results are entered and evaluation during a session, upon saving the results, the e-signature event is raised by the window and the appropriate signoffs take place online. If all the necessary signatures cannot be fulfilled at this time, then the results cannot be committed to the database. Results for all listed tests do not have to be entered in order for the e-signature event to be raised; if one or more test results are entered and saved, then e-signature is required. If more results are entered and evaluated in subsequent sessions, then e-signature is required at that time.
E-record for Results Entry
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.rslt.entry
2. Create AME Rule for the following transaction type:
-
GMD ERES Results Entry
Data on the signoff for results entry includes:
-
Organization, Sample Number, Description, Sample Disposition, Item, Item Revision, Item Description, Grade, Parent Lot, Lot
-
Spec Name, Spec Version
-
Results Summary
Seq, Test, Replicate, Result, Unit, Result Date, Deleted
-
Results Details
Test Sequence, Test, Test Description, Test Class, Test Method, Test Replicate, Result, Test Data Type, Test unit, Result Date, Test Min, Test Max, Test Priority, Display Label, Expression, Lot Optional, Lot Retest, Quality Laboratory, Tester, Reserve Sample, Planned Test Qty, Actual Test Qty, Actual Test UOM, Test Kit Item, Test Kit Lot, Test Provider, Deleted, Spec Target, Spec Min, Spec Max
The following graphic illustrates an example of result entry event.

E-record for Mass Results Entry
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.rslt.mentry
2. Create AME Rule for the following transaction type:
-
GMD ERES Mass Results Entry
Data on the signoff for mass results entry includes:
-
Test, Test Description, Test Class, Test Method, Test Data Type, Expression
-
Samples Used
Organization, Sample Number, Replicate, Result, Test Unit, Result Date, Tester
The following graphic illustrates an example of mass result entry event.

E-record for Results Evaluation
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.rslt.eval
2. Create AME Rule for the following transaction type:
-
GMD ERES Results Evaluation
Data on the signoff for results evaluation includes:
-
Organization, Sample Number, Description, Sample Disposition, Item, Item Revision, Item Description, Grade, Parent Lot, Lot, Spec Name, Spec Version
-
Results Summary
Seq, Test, Replicate, Result, Unit, Result Date, Deleted
-
Results Details
Test Sequence, Test, Test Description, Test Alias, Test Class, Test Method, Test Replicate, Result, Test unit, Test Data Type, Result Date, Parent Sample, Test Min, Test Max, Test Priority, Display Label, Expression, Lot Optional, Lot Retest, In-Spec, Evaluation, Action, Quality Laboratory, Tester, Reserve Sample, Planned Test Qty, Actual Test Qty, Acual Test UOM, Test Kit Item, Test Kit Lot, Test Provider, Deleted, Spec Target, Spec Min, Spec Max
The following graphic illustrates an example of results evaluation event.
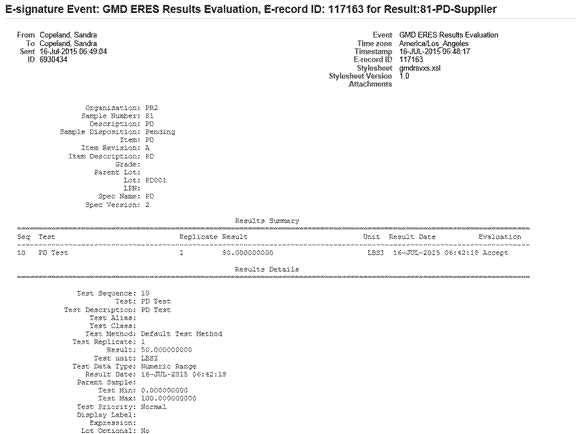
Sample and Sample Group Disposition
The Process Quality Sample Disposition event represents the final review and approval or rejection of all of the results logged against a sample. After the sample disposition is accepted, accepted with variance, or rejected, the lot status is changed to reflect this disposition. Changing to a final sample group disposition can also control the generation of recommended item lot interclass UOM conversions.
The Certificate of Analysis report and Specification Matching feature both depend on a sample group disposition being accepted. If a sample group is not accepted, then these programs do not consider the sample group. Therefore, it is important that the sample group Disposition field is updated.
E-record for Sample Disposition
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.smpl.disp
Create AME Rule for the following transaction type:
-
GMD ERES Sample Disposition
Data on the signoff for sample disposition change includes:
-
Organization, Sample Number, Sample Description, Disposition, Priority
-
Item, Item Revision, Item Description, Source, Source Comments
-
Parent Lot, Lot, Subinventory, Locator
-
License Plate Number (LPN)
-
Customer, Operating Unit, Ship To Location, Order, Order Type, Order Line
-
Batch, Step, Charge, Recipe, Version, Formula, Version, Formula Line, Routing, Version, Operation, Version
-
Internal Orders, Supplier, Supplier Name, Operating Unit, Site, PO Number, PO Line, Receipt Number, Receipt Line, Source Organization, Source Subinventory, Requisition Number, Requisition Line
-
Quality Laboratory, Update Inventory Qty, Sample Qty, Sample Qty UOM
-
Date Drawn, Expiration Date, Samples Instance, Lot Retest Sample, Samples Taken, Storage Subinventory, Storage Locator
-
Spec Name, Spec Version
-
Results Summary
Seq, Test, Replicate, Result, Unit, Result Date, Evaluation
-
Results Details
Test Sequence, Test, Test Description, Test Class, Test Method, Test Replicate, Test Data Type, Result, Test Unit, Result Date, Test Target, Test Min, Test Max, Display Label, Expression, Test Priority, Lot Optional, Lot Retest, LPN, Spec Target, Spec Min, Spec Max, In-Spec, Evaluation, Action, Quality Laboratory, Tester, Planned Test Qty, Actual Test Qty, Test Qty UOM, Test Kit Item, Test Kit Lot, Test Provider
The following graphic illustrates an example of sample disposition event.

E-record for Sample Group Disposition
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.evt.disp
Create AME Rule for the following transaction type:
-
GMD ERES Sample Group Disposition
Data on the signoff for sample group disposition change includes:
-
From Disposition, To Disposition, Item, Item Revision, Item Description, From Grade, To Grade
-
Parent Lot, Lot, Subinventory, Locator
-
License Plate Number (LPN)
-
Customer, Operating Unit, Ship To Location, Order, Order Type, Order Line
-
Batch, Recipe, Version, Formula, Version, Formula Line, Routing, Version, Step, Charge, Operation, Version
-
Internal Orders, Supplier, Supplier Name, Operating Unit, Site, PO Number, PO Line, Receipt Number, Receipt Line, Source Organization, Source Subinventory, Requisition Number, Requisition Line
-
Samples Taken, Samples Required, Samples Type
-
Samples Summary
Sample Number, Disposition, Date Drawn
-
Results Summary
Sample, Seq, Test, Replicate, Result, Unit, Result Date, Evaluation
-
Results Details
Sample, Test Sequence, Test, Test Description, Test Class, Test Method, Test Replicate, Test Priority, Test Data Type, Result, Test Unit, Result Date, Display Label, Expression, In-Spec, Evaluation, Action, Quality Laboratory, Tester, Test Kit Item, Test Kit Lot, Test LPN, Test Provider, Spec Target, Spec Min, Spec Max
The following graphic illustrates an example of sample group disposition event.

E-record for Sample Disposition Composite Change Disposition
To generate ERES event, you must perform the following steps:
1. Enable following event and its subscription:
-
oracle.apps.gmd.qm.evt.dispcom
Create AME Rule for the following transaction type:
-
GMD ERES Sample Group Disposition Composite
Data on the signoff for sample disposition composite change disposition includes:
-
From Disposition, To Disposition, Item, Item Revision, Item Description, From Grade, To Grade
-
Parent Lot, Lot, Subinventory, Locator
-
Customer, Operating Unit, Ship To Location, Order, Order Type, Order Line
-
Batch, Recipe, Version, Formula, Version, Formula Line, Routing, Version, Step, Charge, Operation, Version
-
Internal Orders, Supplier, Supplier Name, Operating Unit, Site, PO Number, PO Line, Receipt Number, Receipt Line, Source Organization, Source Subinventory, Requisition Number, Requisition Line
-
Update Inventory Qty, Control Batch Step Status, Control Lot Attributes, In-Spec Lot Status, Out-of-Spec Lot Status, Hold Date
-
Samples Taken, Samples Required
-
Samples Summary
Org, Sample Number, Disposition, Date Drawn
The following graphic illustrates an example of sample group disposition composite event.

Stability Study Change Status
The stability change status functionality allows you to change stability study status from new to approved, approved to launch, and launch to completion.
Data on the signoff for Stability Study Change Status can include:
-
Organization, Stability Study, Study Description, From Current Status, To Status
-
Item, Item Revision, Item Description, Master Spec, Quality Laboratory, Owner, Notification Lead Time, Testing Grace Period
-
Material Source Count, Storage Condition Count, Package Count
-
Scheduled Start Date, Scheduled End Date, Revised Start Date, Revised End Date, Actual Start Date, Actual End Date
-
Variant Summary
-
Variant Number, Default Item Spec, Default Item Spec Version, Retained Samples
-
-
Variant Material Source
-
Organization, Batch, Recipe, Lot
-
-
Variant Storage Condition
-
Organization, Storage Spec, Storage Spec Version, Resource, Subinventory, Locator, Storage Date
-
-
Variant Package
-
Package, Formula, Formula Version, Sample Quantity/UOM
-
-
Variant Timepoints
-
Time Interval, Spec, Spec Version, Scheduled Date, Actual Date, Samples Required, Disposition, Time Interval, Spec, Spec Version, Scheduled Date, Actual Date, Samples Required, Disposition
-