Update Manufacturer Code List
For Device Products with European licenses, Configuration of Manufacturers and Authorized representative is recommended. This ensures population of Submitter information Sec 1.3 of MIR Report.
Manufacturer code list is used to support configuration of both Manufacturer and Authorized Representatives.
It is recommended that the following fields of Manufacturer codelist are updated to support the mandatory fields of the MIR Report.
- Name
- Contact
- City
- Address
- State/Province
- Postal Code
- Phone
- Country
- Email Address
Note:
The Contact field in the Manufacturer Codelist is used to deriveManufacturer/Authorized Representative Contact's First Name and Contact's Last Name data. It is recommended that first name and last name are configured in a single field with the ‘;‘ delimiter.
Example: John Rich; Configuration of Manufacturers and Authorized Representatives for Existing Products.
- Configure Authorized Representative field and Manufacturer field available in the Product Screen.
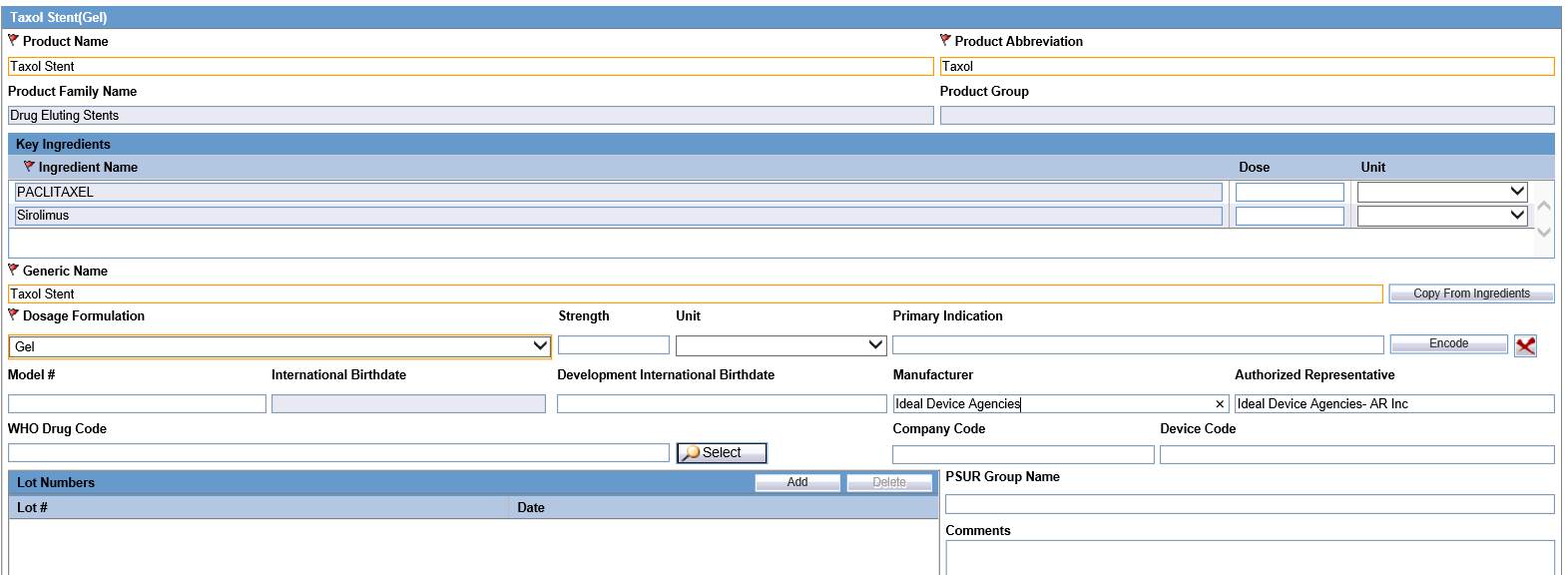
- It is recommended that Device Products have Manufacturer and Authorized Representative fields configured. It is important that if the device product that is manufactured outside Europe, it has a mandatory authorized representative to support Authorized Representative data population.
- The European Commission (EC) recommends that the Authorized Representative must belong to EEA (European Economic Area) Country. Therefore it is recommended that Authorized Representative’s Country is one of the EEA Countries below.
Note:
It is recommended that the EEA Attribute in Countries Flexible Codelist for CH - Switzerland is removed.| Country Code | Country Name |
|---|---|
| AT | Austria |
| BE | Belgium |
| BG | Bulgaria |
| CZ | Czech Republic |
| CY | Cyprus |
| DE | Germany |
| DK | Denmark |
| EE | Estonia |
| ES | Spain |
| FI | Finland |
| FR | France |
| GR | Greece |
| HR | Croatia |
| HU | Hungary |
| IE | Ireland |
| IS | Iceland |
| IT | Italy |
| LI | Liechtenstein |
| LT | Lithuania |
| LU | Luxemburg |
| LV | Latvia |
| MT | Malta |
| NL | Netherlands |
| NO | Norway |
| PL | Poland |
| PT | Portugal |
| RO | Romania |
| SE | Sweden |
| SI | Slovenia |
| SK | Slovakia |
| TR | Turkey |
Note:
Harmonized Codes with IMDRF and FDA Codes for Annexures A to F are preloaded in the repository (CFG_FDA_IMDRF_CODES). Annexure G is preloaded into DEVICE_SUBCOMPONENT flexible code list.Parent topic: Configurations to enable MIR Reporting