Folder structure for multiple studies
To set up several studies for a single sponsor, consider creating a separate folder for each study. PFRInit, the utility that you run when you set up the Reporting and Analysis module for a study, does this automatically.
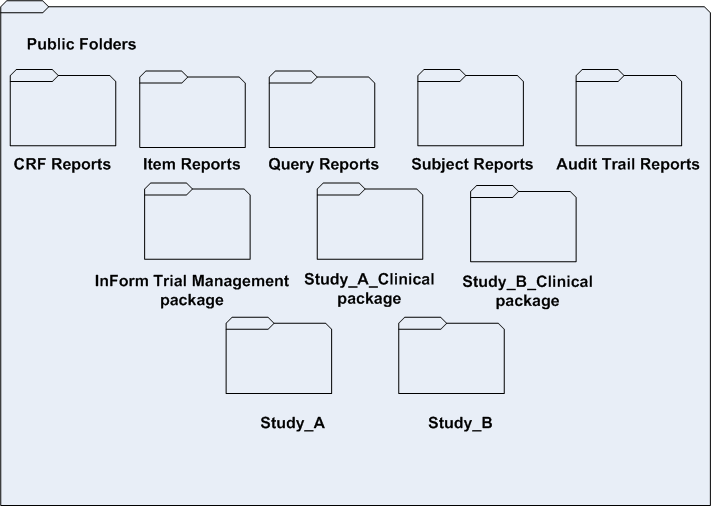
The following illustration shows what the Public Folders portal might contain in the Reporting and Analysis module with folders for two studies (Study A and Study B).
Note that this configuration uses three different packages:
- InForm Trial Management package—Shared by both Study_A and Study_B.
- Study__A_Clinical package—Used only for Study_A.
- Study__B_Clinical package—Used only for Study_B.
The contents of each study-specific folder are set up to include the default reporting folder structure.
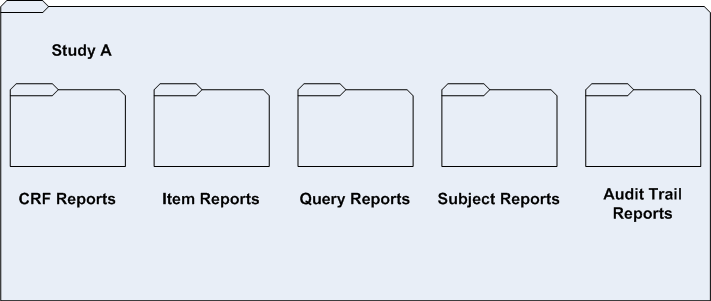
This structure ensures that you can save study-specific properties, such as prompt values and report schedules, for a specified report.



