| Oracle® Health Sciences Clinical Development Analytics User's Guide Release 3.1 E50447-02 |
|
|
PDF · Mobi · ePub |
| Oracle® Health Sciences Clinical Development Analytics User's Guide Release 3.1 E50447-02 |
|
|
PDF · Mobi · ePub |
Using the Oracle BI Analysis Editor, you can run or display predefined reports (delivered with OHSCDA), and you can also create or modify custom analyses.
This chapter contains the following topics:
See Also:
Oracle Fusion Middleware User's Guide for Oracle Business Intelligence Enterprise Edition 11g Release 1 (11.1.1
For ease of reference, the subsequent sections contain excerpts from Oracle Fusion Middleware User's Guide for Oracle Business Intelligence Enterprise Edition 11g Release 1 (11.1.1), altered to include specific information related to OHSCDA.
OHSCDA is delivered with 109 predefined analyses. A subset of these analyses is displayed on your dashboard pages based on the user group you are assigned to.
A analysis can contain data in a tabular format, pivot table, or graph. You can filter the data visible on the analysis based on certain criteria such as Study, Study Site, CRA. These filters are called prompts. Some analyses let you drill down to a more detailed analysis.
Following are some of the analysis-related tasks that you can perform:
Create and modify analyses.
Refresh the results.
Print and Save analyses.
Add analyses to the briefing book.
Copy analyses.
See Also:
Chapter 2, Using Oracle Health Sciences Clinical Development Analytics for more information about analysis-related tasks.
Appendix A, Dashboards and Analyses for more information about predefined analyses.
Oracle Health Sciences Clinical Development Analytics Secure Installation and Configuration Guide for instruction on installing OHSCDA.
Oracle Fusion Middleware User's Guide for Oracle Business Intelligence Enterprise Edition 11g Release 1 (11.1.1)
Perform the following steps to view a predefined analysis:
Log in to OHSCDA.
Using the global header, select the dashboard page that contains the analysis you want to view.
Figure 3-1 displays the CRA dashboard of an administrator.
Note:
Every predefined dashboard page displays the ETL Refresh Date at the bottom of the page. This date indicates the date when the data warehouse was last refreshed.Parameters are displayed if you can filter the analyses. Figure 3-2 displays the parameters in the eCRF Workload page.
Figure 3-2 Parameters Displayed in the CRA Overview Tab of the CRA Dashboard
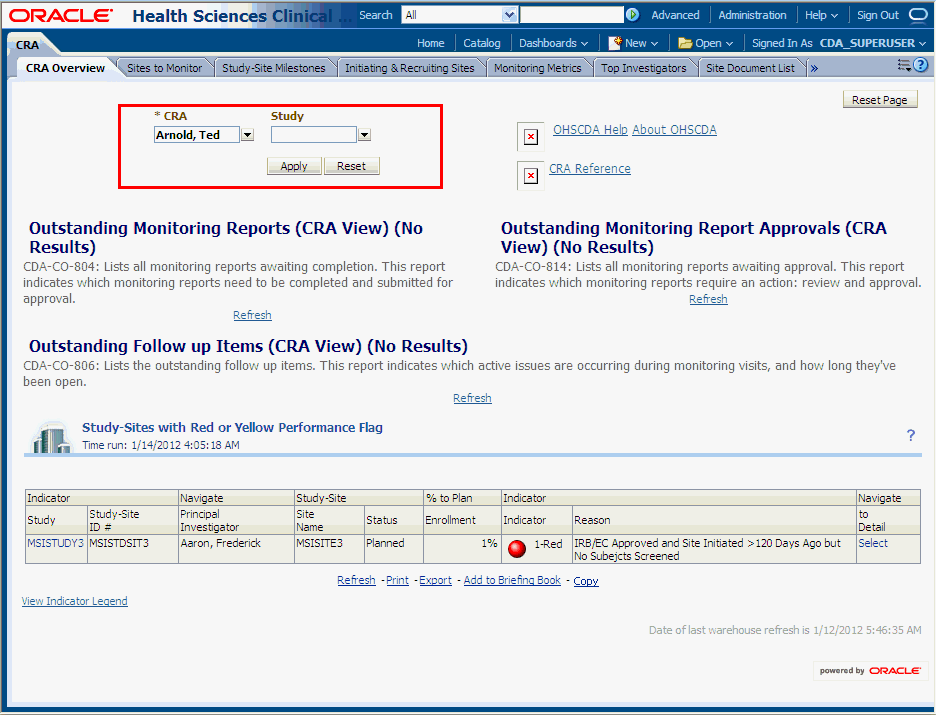
To filter the analysis based on selected criteria:
Select the value from the parameters displayed.
Click Apply.
See Also:
Oracle Fusion Middleware User's Guide for Oracle Business Intelligence Enterprise Edition 11g Release 1 (11.1.1)
You can create a custom analysis if you have been assigned the required permissions and responsibilities. If you prefer, you can copy a pre-defined analysis and use it as a template for the new analysis.
Use the Oracle BI Answers user interface to create your own analysis. But before you proceed, consider the following points:
Do not modify a predefined analysis. If you do, your changes will be overwritten when a new release of OHSCDA is installed. Alternatively, you can make a copy, and modify the copy.
Define a Catalog Folder hierarchy that gives each Answers user a unique folder tree. Limit the right to move analyses into public folders to members of the OHSCDA Administrators group.
Limit the right to place analyses onto shared dashboards to members of the OHSCDA Administrators group.
An action link is a link to an action that you can embed in an analysis that, when clicked, runs an associated action. For more information, refer to Oracle Business Intelligence Presentation Services Administration Guide.
You can create action links for the following columns in OHSCDA 2.0.0.3:
AD05-19 Investigator Action Link
AD07-09 Product Action Link
AD08-06 Site Action Link
AD09-15 Study Action Link
AD10-39 Study-Site Action Link
AD14-08 Team Member Action Link
AD15-19 Contact Action Link
AD16-04 Program Action Link
AD17-04 Region Action Link
TD01-30 Subject Action Link
Oracle Business Intelligence Mobile allows you to view Oracle BI EE content on supported mobile devices such as the Apple iPhone and Apple iPad. Using Oracle Business Intelligence Mobile, you can view and analyze BI content such as analyses and dashboards, BI Publisher content, scorecard content, and content delivered by agents. Oracle Business Intelligence Mobile for Apple iPhone and Apple iPad can be downloaded from the Apple iTunes App Store. Refer to the Oracle® Fusion Middleware User's Guide for Oracle Business Intelligence Enterprise Edition 11g Release 1 (11.1.1) for further information.
The Catalog page lets you locate objects in the catalog and perform tasks specific to those objects. The objects and options that are available to you are determined by your system privileges and the permissions assigned to individual folders and objects. You can get to the Catalog page by clicking Catalog in the Oracle BI global header.
Figure 3-4 shows an example of a Catalog page.
The following table lists the components of the Catalog page:
Table 3-1 Components of the Catalog Page
| Component | Description |
|---|---|
|
Folders Pane |
Displays the catalog as a directory hierarchy or only those folders and objects that you created or marked as your favorites. |
|
Search Pane |
Provides search criteria and initiates a catalog search. The search pane appears by default when you click Advanced on the global toolbar. |
|
Tasks Pane |
Initiate tasks specific to an object. Following are the options in the tasks pane:
|
|
Preview Pane |
Provides a preview of the object that you selected in the Catalog area. This pane is not visible until you click the button to display it. |
|
Location |
Displays the directory path of a selected object. Use as a way to determine where you are within the directory structure. |
|
Show Hidden Items |
When you select this option, hidden folders, are displayed. |
|
Toolbar |
Following are the buttons in the toolbar. The buttons vary between object types.
|
|
Catalog Area |
Workspace that displays the results of your search, opens folders to browse content, and initiates tasks specific to an object. Following are the components of the catalog area:
|
To view saved analyses as stored in the Catalog:
Log in to OHSCDA and click the Catalog link in the global header.
Expand the appropriate folder in the catalog area and select the analysis you want to view.
See Also:
Appendix B, Oracle Health Sciences Clinical Development Analytics Presentation Catalog
The dashboard page lets you view and navigate a dashboard. Figure 3-9 shows an example of a dashboard page.
Following are the components of a dashboard page:
Page Tabs: Use the tabs across the top of the viewer to select the dashboard page to view and navigate.
Toolbar: Contains the Page Options button, which displays the following options:
Note:
You might not see some of the options depending on you user privileges.Edit Dashboard — Use this button to display the Dashboard builder, where you modify the dashboard, if you have the appropriate permissions.
Print — Use this option to print the current dashboard page as either HTML or PDF.
Refresh — Use this option to refresh the results of analyses included in the dashboard page.
Add to Briefing Book — Use this option to add the contents of a dashboard page to a briefing book. (This option is not available on an empty dashboard page or if the Show Add to Briefing Book box in the "Dashboard Properties dialog", has not been selected for the page.)
Create Bookmark Link — Use this option to create a bookmark link.
Create Prompted Link — Use this option to create a prompted link.
Apply Saved Customization — Use this option to apply a saved customization — either a personal saved customization or a shared saved customization.
Save Current Customization — Use this option to save your current customization.
Edit Saved Customizations — Displays the Edit Saved Customizations dialog where you edit details of your saved customizations.
Clear My Customization — Use this option to clear your current customization.
To view saved analyses organized by dashboard:
Log in to OHSCDA and click Dashboard in the global header. A drop-down of saved analyses appears.
Select the analysis you wish to view.
This section describes the indicators in OHSCDA 2.2.
The following section describes indicators for studies.
The Study Key Performance Indicator (KPI) Flag provides a single red, yellow or green icon, which indicates that one or more study-level KPIs is at risk of missing a target (yellow) or has missed a target (red). If none of the KPIs is red or yellow, the Study KPI Flag shows green.
The Study KPI Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. Consequently, it only evaluates cycles that have not yet completed. For example, Protocol Approved to 1st Subject Enrolled is only evaluated if the 1st Subject has not yet been enrolled.
The Study KPI Flag evaluates the following KPIs:
Last Subject Last Visit to Database Lock
Protocol Approved to 1st Subject Enrolled
Protocol Approved to 1st Site Initiated
Each KPI is evaluated based on the following rules:
Last Subject Last Visit (LSLV) to Database (DB) Lock calculation is evaluated when LSLV is complete but DB Lock has not yet been reached.
KPI at risk of missing target. The Target cycle time will be reached in the next 30 days.
High Level Reason - Missed KPI (s)
Detail Reason - Missed KPI: Last Subject Last Visit to Database Lock
KPI has missed target. The time since LSLV is greater than the Target cycle time.
High Level Reason - KPI(s) at Risk
Detail Reason - KPI(s) at Risk (30 days): Last Subject Last Visit to Database Lock
Protocol Approved to 1st Subject Enrolled is evaluated when protocol has been approved but 1st subject has not yet been enrolled.
KPI has missed target. The time since protocol approved is greater than the Target cycle time.
High Level Reason - Missed KPI (s)
Detail Reason - Missed KPI: Protocol Approved to 1st Subject Enrolled
KPI at risk of missing target. The Target cycle time will be reached in the next 30 days.
High Level Reason - KPI(s) at Risk
Detail Reason - KPI(s) at Risk (30 days): Protocol Approved to 1st Subject Enrolled
Protocol Approved to 1st Site Initiated is evaluated when protocol has been approved but 1st site has not yet been initiated. If protocol has been approved and neither has the 1st site been initiated nor 1st subject enrolled, the system evaluates Protocol Approved to 1st Subject Enrolled before evaluating Protocol Approved to 1st Site Initiated.
KPI has missed target. The time since protocol approved is greater than the Target cycle time.
High Level Reason - Missed KPI (s)
Detail Reason - Missed KPI: Protocol Approved to 1st Site Initiated
KPI at risk of missing target. The Target cycle time will be reached in the next 30 days.
High Level Reason - KPI(s) at Risk
Detail Reason - KPI(s) at Risk (30 days): Protocol Approved to 1st Site Initiated
The Study Milestone Flag provides a single red, yellow or green icon, which indicates that one or more study-level milestones is at risk of missing a planned/latest estimate (LE) date (yellow) or has missed a planned/LE date (red). If none of the milestones is red or yellow, the Study Milestone Flag shows green.
All dates evaluate the LE date, if populated. If the LE date is not populated, the planned date is used. If neither the Planned nor LE dates is populated, the milestone is not evaluated.
The Study Milestone Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. Consequently, it only evaluates milestones where a planned or LE date exists but an actual date has not yet been entered.
The Study Milestone Flag evaluates the following milestones:
Protocol Approved Date
1st Site Initiated Date
1st Critical Document Package Completed Date
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
Database Lock Date
Clinical Study Report Date
Each milestone is evaluated based on the following rules:
Protocol Approved Date is evaluated only when none of the following post-protocol approval activities have occurred (Actual date is null):
1st Site Initiated Date
1st Critical Document Package Completed Date
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
Database Lock Date
Clinical Study Report Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Protocol Approved Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Protocol Approved Date
1st Site Initiated Date is evaluated only when none of the following activities have occurred (Actual date is null):
1st Critical Document Package Completed Date
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
Database Lock Date
Clinical Study Report Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: 1st Site Initiated Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: 1st Site Initiated Date
1st Critical Document Package Completed Date is evaluated only when none of the following activities have occurred (Actual date is null):
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
Database Lock Date
Clinical Study Report Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: 1st Critical Document Package Completed Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: 1st Critical Document Package Completed Date
Last Subject Enrolled Date is evaluated only when none of the following activities have occurred (Actual date is null):
Last Subject Last Visit Date
Database Lock Date
Clinical Study Report Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Enrolled Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Enrolled Date
Last Subject Last Visit Date is evaluated only when none of the following activities have occurred (Actual date is null):
Database Lock Date
Clinical Study Report Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Last Visit Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Last Visit Date
Database Lock Date is evaluated only when none of the following activities have occurred (Actual date is null):
Clinical Study Report Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Last Visit Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Last Visit Date
Last Subject Last Visit Date is evaluated only when none of the following activities have occurred (Actual date is null):
Database Lock Date
Clinical Study Report Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Last Visit Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Last Visit Date
The Study Recruitment Flag provides a single red, yellow or green icon, which indicates whether recruitment is progressing normally. The Study Recruitment Flag is set up to evaluate three different scenarios:
Enrollment Delay - The enrollment delay flag is triggered when there hasn't been an enrollment within 120 days (red) or 90 days (yellow) of 1st Site Initiated.
Screen Failure Rate - The screen failure rate flag is triggered when the screen fail rate is 20% or more (red) or 10-20% more than the expected screen failure rate for the study.
Enrollment Lag - The enrollment lag flag is triggered based on how much of the planned enrollment period is remaining, using Planned / LE and Actual 1st Subject Enrolled Date, and Planned / LE Last Subject Enrolled Date:
Late Enrollment (25% or less of planned enrollment period remaining) - triggered when less than 100% of planned subjects have been enrolled and it has been more than 30 days (red) or 15-30 days (yellow) since the last enrollment.
Mid Enrollment (25-50% of planned enrollment period remaining) - triggered when less than 100% of planned subjects have been enrolled and it has been more than 45 days (red) or 30-45 days (yellow) since the last enrollment.
Early Enrollment (50% or more of planned enrollment period remaining) - triggered when less than 100% of planned subjects have been enrolled and it has been more than 60 days (red) or 45-60 days (yellow) since the last enrollment.
If any one of the scenarios above is red, the Study Recruitment Flag is red. If none are red and one or more is yellow, the Study Recruitment Flag is yellow. If none is red or yellow, the Study Recruitment Flag shows green.
The Study Recruitment Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates recruitment if the Last Subject Enrolled Date and Database Lock Dates have not yet been populated.
Each milestone is evaluated based on the following rules:
120 days or more have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Enrollment Delay
Detail Reason - 1st Site Initiated > 120 Days Ago but no 1st Subject Enrolled
90-120 days have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Enrollment Delay
Detail Reason - 1st Site Initiated > 90 Days Ago but no 1st Subject Enrolled
120 days or more have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Screen Fail Rate
Detail Reason - Screen Fail Rate > 20% Variance from Expected for Study
90-120 days have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Screen Fail Rate
Detail Reason - Fail Rate between 10-20% Variance from Expected for Study
Less than 100% planned enrollment completed, 30 days or more have passed since last enrollment and 25% or less of enrollment period remains.
High Level Reason - Enrollment Lag
Detail Reason - 30 Days Since Last Enrollment and Less than 25% of Enrollment Period Remaining (includes % planned enrollment completed)
Less than 100% planned enrollment completed, 15-30 days have passed since last enrollment and 25% or less of enrollment period remains.
High Level Reason - Enrollment Lag
Detail Reason - 15 Days Since Last Enrollment and Less than 25% of Enrollment Period Remaining (includes % planned enrollment completed)
Less than 100% planned enrollment completed, 45 days or more have passed since last enrollment and 25-50% of enrollment period remains.
High Level Reason - Enrollment Lag
Detail Reason - 45 Days Since Last Enrollment and 25-50% of Enrollment Period Remaining (includes % planned enrollment completed)
Less than 100% planned enrollment completed, 30 days have passed since last enrollment and 25-50% of enrollment period remains.
High Level Reason - Enrollment Lag
Detail Reason - 30 Days Since Last Enrollment and 25-50% of Enrollment Period Remaining (includes % planned enrollment completed)
Less than 100% planned enrollment completed, 60 days or more have passed since last enrollment and more than 50% of enrollment period remains.
High Level Reason - Enrollment Lag
Detail Reason - 60 Days Since Last Enrollment and more than 50% of Enrollment Period Remaining (includes % planned enrollment completed)
Less than 100% planned enrollment completed, 45-60 days have passed since last enrollment and more than 50% of enrollment period remains.
High Level Reason - Enrollment Lag
Detail Reason - 45 Days Since Last Enrollment and more than 50% of Enrollment Period Remaining (includes % planned enrollment completed)
The Study Retention Flag provides a single red, yellow or green icon, which indicates that the early termination rate for the study is more than 20% (red) or 1-20% more than the expected early termination rate. If the actual early termination rate for the study is less than or equal to the expected rate, the Study Retention Flag shows green.
The Study Retention Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates studies that have an actual early term rate and where 25% or more of planned subjects have been enrolled.
Study Retention is evaluated when % Subjects Early Terminated is not null and where % Planned Subjects Enrolled in the study is greater than or equal to 25%.
Actual early termination rate is 20% or more over expected early termination rate for the study.
High Level Reason - Early Term Rate
Detail Reason - Early Termination Rate > 20% over Expected for Study
Actual early termination rate is between 1-20% over expected early termination rate for the study.
High Level Reason - Early Term Rate
Detail Reason - Early Termination Rate between 1-20% over Expected for Study
The following section describes indicators for study-regions.
The Study-Region Milestone Flag provides a single red, yellow or green icon, which indicates that one or more study-region-level milestones is at risk of missing a planned/latest estimate (LE) date (yellow) or has missed a planned/LE date (red). If none of the milestones is red or yellow, the Study-Region Milestone Flag shows green.
All dates evaluate the LE date, if populated. If the LE date is not populated, the planned date is used. If neither the Planned nor LE dates are populated, the milestone is not evaluated.
The Study-Region Milestone Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates milestones where a planned or LE date exists but an actual date has not yet been entered.
The Study Milestone Flag evaluates the following milestones:
1st Site Initiated Date
1st Critical Document Package Completed Date
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
Each milestone is evaluated based on the following rules:
1st Site Initiated Date is evaluated only when none of the following activities have occurred (Actual date is null):
1st Critical Document Package Completed Date
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: 1st Site Initiated Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: 1st Site Initiated Date
1st Critical Document Package Completed Date is evaluated only when none of the following activities have occurred (Actual date is null):
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: 1st Critical Document Package Completed Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: 1st Critical Document Package Completed Date
Last Subject Enrolled Date is evaluated only when none of the following activities have occurred (Actual date is null):
Last Subject Last Visit Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Enrolled Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Enrolled Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Last Visit Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Last Visit Date
The Study-Region Recruitment Flag provides a single red, yellow or green icon, which indicates whether recruitment is progressing normally. The Study Recruitment Flag is set up to evaluate three different scenarios:
Screening Delay - The screening delay flag is triggered when there hasn't been a first subject screened within 90 days (red) or 60 days (yellow) of 1st Site Initiated for this study-region.
Enrollment Delay - The enrollment delay flag is triggered when there hasn't been an enrollment within 120 days (red) or 90 days (yellow) of 1st Site Initiated for this study-region.
Screen Failure Rate - The screen failure rate flag is triggered when the screen fail rate for this study-region is 20% or more (red) or 10-20% more than the expected screen failure rate for the study.
Enrollment Lag - The enrollment lag flag is triggered when there is at least one subject enrolled, there is less than 50% planned enrollment completed, and it's been more than 60 days (red) or 90 days (yellow) since the last enrollment for this study-region.
If any one of the scenarios above is red, the Study Recruitment Flag is red. If none are red and one or more is yellow, the Study Recruitment Flag is yellow. If none is red or yellow, the Study Recruitment Flag shows green.
The Study Recruitment Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates recruitment if the Last Subject Enrolled Date and Database Lock Dates have not yet been populated.
Each milestone is evaluated based on the following rules:
120 days or more have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Enrollment Delay
Detail Reason - 1st Site Initiated > 120 Days Ago but no 1st Subject Enrolled
90-120 days have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Enrollment Delay
Detail Reason - 1st Site Initiated > 90 Days Ago but no 1st Subject Enrolled
120 days or more have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Screen Fail Rate
Detail Reason - Screen Fail Rate > 20% Variance from Expected for Study
90-120 days have passed since 1st Site Initiated and there are no subjects enrolled.
High Level Reason - Screen Fail Rate
Detail Reason - Fail Rate between 10-20% Variance from Expected for Study
Site enrollment is less than 50% and more than 60 days have passed since last enrollment.
High Level Reason - Enrollment Lag
Detail Reason - Site Enrollment Under 50% and > 60 Days Since Last Enrollment
Site enrollment is less than 50% and 30-60 days have passed since last enrollment.
High Level Reason - Enrollment Lag
Detail Reason - Site Enrollment Under 50% and > 30 Days Since Last Enrollment
The Study-Region Retention Flag provides a single red, yellow or green icon, which indicates that the early termination rate for the study-region is more than 20% (red) or 1-20% more than the expected early termination rate for the study. If the actual early termination rate for the study is less than or equal to the expected rate, the Study-Region Retention Flag shows green.
The Study-Region Retention Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates study-regions that have an actual early term rate and where 25% or more of planned subjects have been enrolled.
Study-Region Retention is evaluated when % Subjects Early Terminated is not null and where % Planned Subjects Enrolled in the study-region is greater than or equal to 25%.
Actual early termination rate is 20% or more over expected early termination rate for the study.
High Level Reason - Early Term Rate
Detail Reason - Early Termination Rate > 20% over Expected for Study
Actual early termination rate is between 1-20% over expected early termination rate for the study.
High Level Reason - Early Term Rate
Detail Reason - Early Termination Rate between 1-20% over Expected for Study
The following section describes indicators for study-sites.
The Study-Site Milestone Flag provides a single red, yellow or green icon, which indicates that one or more study-site-level milestones is at risk of missing a planned/latest estimate (LE) date (yellow) or has missed a planned/LE date (red). If none of the milestones is red or yellow, the Study-Site Milestone Flag shows green.
All dates evaluate the LE date, if populated. If the LE date is not populated, the planned date is used. If neither the Planned nor LE dates are populated, the milestone is not evaluated.
The Study-Site Milestone Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates milestones where a planned or LE date exists but an actual date has not yet been entered.
The Study Milestone Flag evaluates the following milestones:
Site Initiated Date
Critical Document Package Completed Date
1st Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
Each milestone is evaluated based on the following rules:
Site Initiated Date is evaluated only when none of the following activities have occurred (Actual date is null):
Critical Document Package Completed Date
Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Site Initiated Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Site Initiated Date
Critical Document Package Completed Date is evaluated only when none of the following activities have occurred (Actual date is null):
Subject Enrolled Date
Last Subject Enrolled Date
Last Subject Last Visit Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Critical Document Package Completed Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Critical Document Package Completed Date
Last Subject Enrolled Date is evaluated only when none of the following activities have occurred (Actual date is null):
Last Subject Last Visit Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Enrolled Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Enrolled Date
The Planned or LE date is in the past.
High Level Reason - Missed Milestone(s)
Detail Reason - Missed Milestone: Last Subject Last Visit Date
The Planned or LE Date is in the next 30 days.
High Level Reason - Milestone at Risk
Detail Reason - Milestone at Risk: Last Subject Last Visit Date
The Study-Site Recruitment Flag provides a single red, yellow or green icon, which indicates whether recruitment is progressing normally. The Study Recruitment Flag is set up to evaluate four different scenarios:
Screening Delay - The screening delay flag is triggered when there hasn't been a first subject screened within 90 days (red) or 60 days (yellow) of Site Initiated for this study-site.
Enrollment Delay - The enrollment delay flag is triggered when there hasn't been an enrollment within 120 days (red) or 90 days (yellow) of Site Initiated for this study-site.
Screen Failure Rate - The screen failure rate flag is triggered when the screen fail rate for this study-site is 20% or more (red) or 10-20% more than the expected screen failure rate for the study.
Enrollment Lag - The enrollment lag flag is triggered when there is at least one subject enrolled, there is less than 50% planned enrollment completed, and it's been more than 60 days (red) or 90 days (yellow) since the last enrollment for this study-site.
If any one of the scenarios above is red, the Study Recruitment Flag is red. If none are red and one or more is yellow, the Study Recruitment Flag is yellow. If none is red or yellow, the Study Recruitment Flag shows green.
The Study Recruitment Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates recruitment if the Last Subject Enrolled Date and Database Lock Dates have not yet been populated.
Each milestone is evaluated based on the following rules:
120 days or more have passed since Site Initiated and there are no subjects enrolled.
High Level Reason - Enrollment Delay
Detail Reason - 1st Site Initiated > 120 Days Ago but no 1st Subject Enrolled
90-120 days have passed since Site Initiated and there are no subjects enrolled.
High Level Reason - Enrollment Delay
Detail Reason - 1st Site Initiated > 90 Days Ago but no 1st Subject Enrolled
120 days or more have passed since Site Initiated and there are no subjects enrolled.
High Level Reason - Screen Fail Rate
Detail Reason - Screen Fail Rate > 20% Variance from Expected for Study
90-120 days have passed since Site Initiated and there are no subjects enrolled.
High Level Reason - Screen Fail Rate
Detail Reason - Fail Rate between 10-20% Variance from Expected for Study
Site enrollment is less than 50% and more than 60 days have passed since last enrollment.
High Level Reason - Enrollment Lag
Detail Reason - Site Enrollment Under 50% and > 60 Days Since Last Enrollment
Site enrollment is less than 50% and 30-60 days have passed since last enrollment.
High Level Reason - Enrollment Lag
Detail Reason - Site Enrollment Under 50% and > 30 Days Since Last Enrollment
The Study-Site Retention Flag provides a single red, yellow or green icon, which indicates that the early termination rate for the study-site is more than 20% (red) or 1-20% more than the expected early termination rate for the study. If the actual early termination rate for the study is less than or equal to the expected rate, the Study-Site Retention Flag shows green.
The Study-Site Retention Flag is intended for use as an actionable measure, identifying situations where you may be able to affect the outcome in some way. As a result, it only evaluates study-sites that have an actual early term rate and where 25% or more of planned subjects have been enrolled.
Study-Site Retention is evaluated when % Subjects Early Terminated is not null and where % Planned Subjects Enrolled in the study-site is greater than or equal to 25%.
Actual early termination rate is 20% or more over expected early termination rate for the study.
High Level Reason - Early Term Rate
Detail Reason - Early Termination Rate > 20% over Expected for Study
Actual early termination rate is between 1-20% over expected early termination rate for the study.
High Level Reason - Early Term Rate
Detail Reason - Early Termination Rate between 1-20% over Expected for Study