|
|
| HDR Version 7 HL7 Version 3 Messaging Conformance Specification |
| Contents Previous Next Conformance Examples |
This section contains the following topics:
5.72.1 Introduction 5.72.2 RMIM 5.72.3 Referenced CMETs 5.72.4 Links to Artifacts
The R_Medication (universal) CMET captures drug information that is relevant for drug related adverse event and product reporting. This includes information about the manufacturer, distributor, regulatory authority, ingredients, and generic equivalents.
The following figure shows the R_Medication (universal) RMIM:
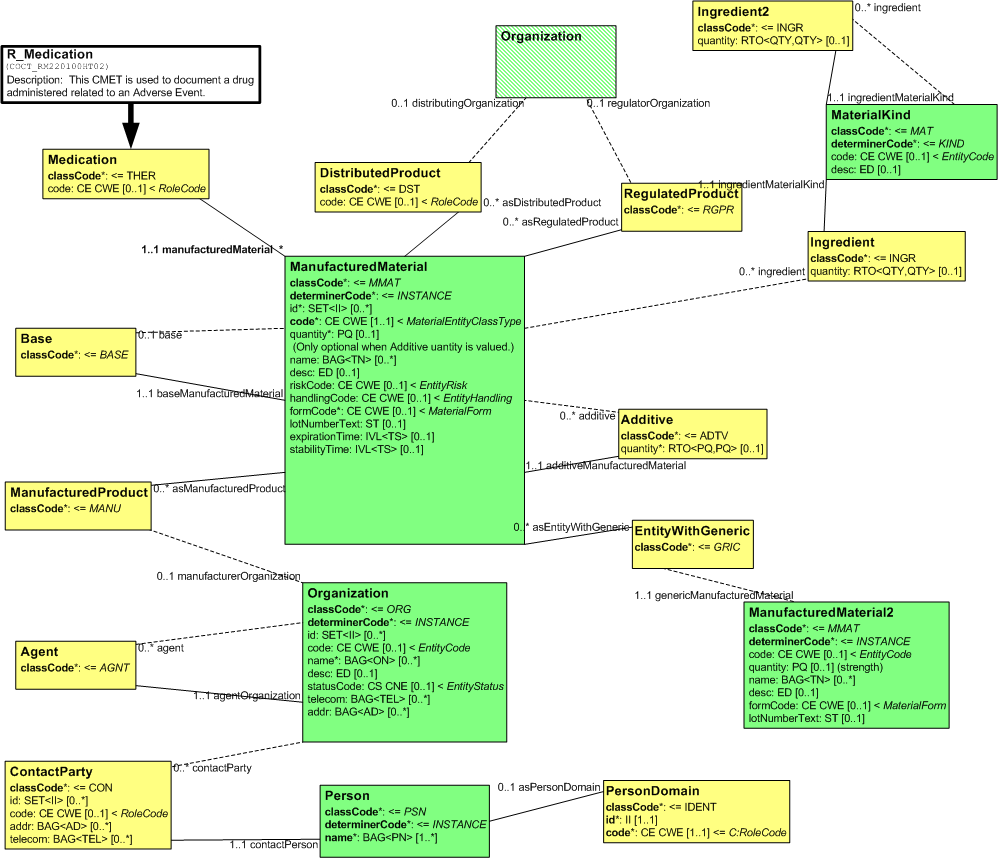
Figure: 5.72.2-1 R_Medication (universal) RMIM
| Artifacts | Link |
| Visio File | coct_rm220100ht02.vsd |
| Visio XML File | COCT_MT220100HT02.xml |
| Hierarchical Message Description (HMD) | |
| Schema | coct_mt220100ht02.xsd |
| Model Interchange Files (MIF) | coct_mt220100ht02.mif |
| Contents Previous Next Conformance Examples |
|
Copyright © 2018, Oracle. All rights reserved. |
About Oracle Contact Us Legal Notices and Terms for Use Privacy Statement |